Algal toxins can cause diarrhoea, vomit, tingling, paralysis and other effects in humans, mammals or fish. Algal toxins are produced by various algae and are found both in seawater and fresh water. The algal toxins can be retained in shellfish or contaminate drinking water. They have no taste or smell, and are not eliminated by cooking or freezing.
A series of such marine algal toxins are known, and among them are okadaic acid and the dinophysistoxins, saxitoxins, brevetoxins, domoic acid, azaspiracids, pinnatoxins, yessotoxins, pectenotoxins and cyclic imines, and most of them are found all over the world.
Azaspiracid
Azaspiracid (AZA) and Azaspiracid shellfish poisoning (AZP) was discovered in November 1995, when at least eight people in the Netherlands became ill after eating mussels (Mytilus edulis) harvested at Killary Harbor, Ireland. In November 1997, the toxicity recurred in mussels from Aranmore Island, Ireland and new human intoxications were reported. Since then AZAs have been found and reported in shellfish from Norway, UK, France, Morocco, Canada and Chile.
Symptoms of AZP
Human symptoms of AZP-poisoning are nausea, vomiting, severe diarrhea and stomach cramps. The symptoms are similar to those of diarrhetic shellfish poisoning (DSP).
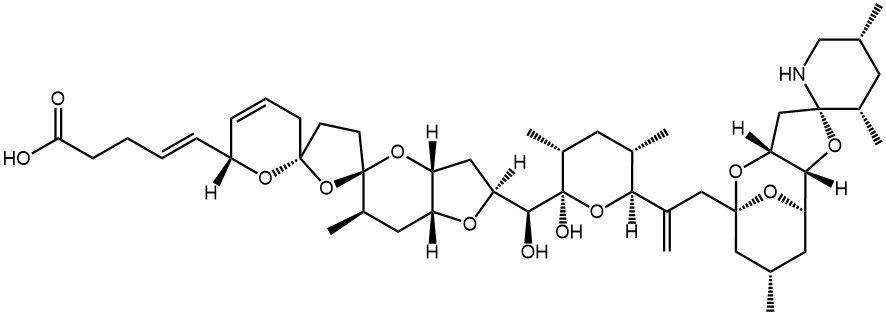
Structure
Azaspiracids have two unique spiro ring asemblies, a cyclic amine and a carboxylic acid, and differs from any of the previously known nitrogen-containing toxins found in shellfish or dinoflagellates. A series of analogues have been identified, whereas AZA-1, AZA-2, AZA-3 and AZA-6 are the most commonly found in shellfish. Today more than 24 analogues of azaspiracids are known.
Okadaic & DTX
Okadaic acid & Dinophysis toxins
Okadic acid (OA) is together with the dinophysis toxins (DTX-1, DTX-2, and DTX-3), one of the most widely common classes of marine biotoxins. The toxins are lipophilic and thermostable polyethers found all over the world. OA and DTXs accumlate in shellfish during blooms of the source algae.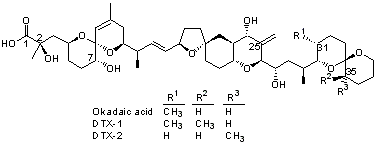
Source organism
Okadaic acid and Dinophysis toxins are compounds produced by Dinophysis species (Lee et al., 1989; Yasumoto et al., 1980) and Prorocentrum lima (Murakami et al., 1982).
Symptoms
The dinophysis toxins causes diarrhetic shellfish poisoning (DSP) with the symptoms diarrhea, nausea, vomiting, abdominal pain and general discomfort (Aune and Yndestad, 1993; van Egmond et al., 1993) appearing within 0.5-6 hours and recovery is usually complete within three days. No deaths have been reported.
Mechanism of action
OA and DTXs act by inhibiting protein phosphatases PP1 and PP2A (Cohen, 1989; Cohen and Cohen, 1989; Haystead et al., 1989). Phosphorylation and dephosphorylation of proteins is one of the major regulatory processes in eukaryotic cells, associated with metabolism, membrane transport and secretion, contractility, cell division and others (Cohen, 1989; Yamashita et al., 1990). It is suggested that diarrhea in man is caused by hyperphosphorylation of proteins that control sodium secretion by intestinal cells (Cohen et al., 1990) or by increased phosphorylation of cytoskeletal or junctional moieties that regulate solute permeability, resulting in passive loss of fluids (Dho et al., 1990).
Yessotoxin
Yessotoxin
Yessotoxin (YTX) was originally isolated in Japan in 1986, from the digestive organ of the scallop Patinopecten yessoensis, from which the name yessotoxin was derived. A year later, YTX was found and reported occurring in Norwegian blue mussels, Mytilus edulis, collected in 1985 and 1986 from Flødevigen and Sogndal. Since then, YTX has been detected in shellfish from New Zealand (1997), Italy (1997), and Chile (1997).
Structure
Yessotoxin is a disulfated polyether compound, consisting of 11 contiguously transfused ether rings (47 carbons), an unsaturated side chain (9 carbons), and two sulphate esters.
Other analogues
Besides YTX, various yessotoxin analogues (YTXs) have been isolated and described, and the first naturally occurring YTX analogue was identified in the Japanese scallop P. yessoensis as 45-hydroxyYTX. Some years later, 45,46,47-trinorYTX was identified in the same shellfish species. In 1998, 1-desulfoYTX was isolated, characterized and reported from Norwegian blue mussels. Since then, a series of new analogues have been described in mussels, e.g. carboxyYTX, homoYTX and 45-hydroxyhomoYTX, adriatoxin, carboxyhomoYTX, 42,43,44,45,46,47,55-heptanor-41-oxohomoYTX, 42,43,44,45,46,47,55-heptanor-41-oxoYTX.
Source organism
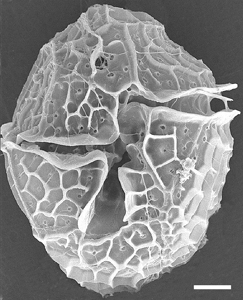

The biogenic source organism for YTX was identified and confirmed by establishing and studying axenic unialgal cultures of a New Zealand strain of Protoceratium reticulatum (= Gonyaulax grindleyi). Production of yessotoxin has since been demonstrated in P. reticulatum isolates from Japan, Italy, Scotland, Canada, Spain and the USA. Lingulodinium polyedrum has also been associated with production of YTXs. Our selves, we have showed P. reticulatum to be a producer of YTXs by analysing single picked cells by YTX-ELISA.
Figure 1. Light and scanning electron microscopy of P. reticulatum. Left photo: Nomarski interference contrast microscopy of a living cell. Right photo: Scanning electron microscopy of CalcoFluor white stained cell. Scale bars: 5 µm. (Johannsen, 2004).
Classification
YTX was initially identified because of its interference with the mouse bioassay. This interference led to the initial classification of YTX as a Diaretic Shellfish Poisoning (DSP) toxin. Since, diarrhea has not been observed by either i.p. or oral administration of YTXs, the appropriateness of classifying these compounds as DSP toxins is questionable. This is supported by a study showing that YTX is not a protein phosphatase inhibitor. Recent recommendations are that YTXs should no longer be included in the DSP class, but rather considered as a separate class of toxins.
Toxicology and human relevance
Several studies have been conducted to assess the potential toxicity of YTXs. To date none of these studies have highlighted any toxic effects of YTXs in regards to humans. They have however found that YTXs have toxic effects in mice when the YTX had been administered by an intraperitoneal injection into the animal. The toxicicological effects encountered are similar to those seen for paralytic shellfish toxins and include hepatotoxicity, cardiotoxicity and neurotoxicity, with a YTX level of 80-750 μg/kg causing toxic effects. Limited toxic effects have been seen after oral administration of the toxin to animals, even with 54 mg/kg of YTX given to one mouse. The mechanism of action is unknown and is currently being studied by a number of research groups.
Pinnatoxin
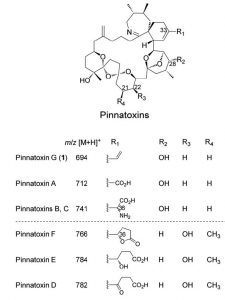 Pinnatoxins
Pinnatoxins
Pectenotoxin
Pectenotoxins (PTXs) are a group of toxins originating from Dinophysis species throughout the world. PTX-2 has been identified in Dinophysis fortii from Japan and Italy and Dinophysis acuta from New Zealand and Ireland.
Spirolides
Spirolides